Modern periodic table is the systematic arrangement of elements based on increasing atomic number. The formation of this periodic table is not a matter of months or years. It took centuries to evolve.
History of Periodic Table
From the beginning of human history till 17th century, humans knew only a dozen of elements like gold, iron and copper etc. With the progress of science and instruments, more elements were discovered. The list of elements became so long that it was difficult to study them without a proper system. To solve this problem, different chemists presented different laws to arrange elements in systematic ways so that their study could be made easier. In 19th century, different chemists tried to identify elements with similar properties and group them together as follows:
1. Antoine Lavoisier
In 1789, the French chemist Antoine Lavoisier was the first to classify elements into metals and non-metals. He included 30 substances in his table of elements. However, he was confused because he also considered light and heat as elements.
2. Dobereiner’s Triads
In 1829, a German chemist, Johann Wolfgang Dobereiner (1780-1849) put forward his famous law called Dobereiner’s law of triads. He made groups of different elements that shared similar properties. Each group contained three elements, which he called triads (tri-means three). He stated that in each group, the atomic mass of the second element of triad was average of the atomic masses of the first and third element) as shown below.
| Triad A | Elements | Lithium | Sodium | Potassium |
| Atomic Weights | 6.94 | 23.02 | 39.10 | |
| Triad B | Elements | Calcium | Strontium | Barium |
| Atomic Weights | 40.1 | 87.6 | 137.3 | |
| Triad C | Elements | Chlorine | Bromine | Iodine |
| Atomic Weights | 35.4 | 79.9 | 126.9 | |
| Triad D | Elements | Sulphur | Selenium | Tellurium |
| Atomic Weights | 32.1 | 78.9 | 127.6 | |
| Triad E | Elements | Iron | Cobalt | Nickel |
| Atomic Weights | 55.8 | 58.9 | 58.7 |
By looking at Triad A, you will find that the atomic mass of second element (sodium) is 23.02 amu which is nearly average of the atomic masses of the first and second elements, lithium (6.94 amu) and potassium (39.10 amu). All three elements share similar properties. Even today modern periodic table places them in same group (Alkali metals).
Similarly, in Triad C has chlorine, bromine and iodine having similar chemical properties as evident from modern periodic table. The atomic mass of second element (bromine) is average of the first element (chlorine) and third element (iodine). The same pattern is observed throughout the table following Dobereiner model. Dobereiner’s law of Triads could not be applied to all elements of that times, however, this law laid foundation for further research and development of modern periodic
3. Newland’s Law of Octaves
Döbereiner’s law of triads failed because it could not be applied to all the elements known at that time. An English chemist, John Alexander Newlands (1838–1898), attempted to include all elements in his arrangement. He arranged the elements in order of increasing atomic masses and designed a table in which every eighth element showed properties similar to the first. He called this relationship the Law of Octaves (from octa, meaning eight). The table below shows the arrangement of elements given by Newlands.
| Element | Li | Be | B | C | N | O | F |
| Atomic Weight | 7 | 9 | 11 | 12 | 14 | 16 | 19 |
| Element | Na | Mg | Al | Si | P | S | Cl |
| Atomic Weight | 23 | 24 | 27 | 29 | 31 | 32 | 35.5 |
| Element | K | Ca | |||||
| Atomic Weight | 39 | 40 |
The Newland’s Law of Octaves also failed because that could only be applied upto calcium element. However, his work would always be appreciated in the history of development of period table of elements.
4. Lothar Meyer’s Periodic Table
In 1869, a German chemist named Lothar Meyer (1830-1895) arranged 28 elements according to their increasing atomic weights and found that they showed similarity in their properties after regular intervals. He examined that many physical properties of elements (boiling points and hardness etc) change in regular pattern. This means that those properties of elements are repeated after regular intervals of time with increasing atomic masses. He presented his work in the form of a graph plotted between atomic volume and atomic mass and published in 1870.
He wasted time in getting his work published because he was reluctant to whether keep some spaces for the elements to discover in future or not. Otherwise, he had made two periodic tables, one in 1864 and the other in 1869 (some months before Dmitri Medndeleev’s periodic table)
5. Dmitri Mendeleev’s Periodic Table
A Russian chemist named Dmitri Mendeleev (1834-1907), was busy in writing a textbook for chemistry students. He wanted to arrange elements in his textbook to study them easily. In doing so, he organized elements on a paper in increasing atomic masses and found that their properties repeated after regular intervals.
In 1869, Dmitri Mendeleev published his periodic table, one year before Lothar Meyer’s periodic table.
His work is significant compared to other chemists because he knew that not all elements had been discovered, only 63 elements were discovered at that time. Based on similarity (repetition) of properties of elements with increasing atomic masses after regular intervals, he predicted the properties of those undiscovered elements and left blank spaces for them in his periodic table. For example, he left spaces at atomic masses 44, 68, 72, and 100, predicting that elements with these masses would be discovered in the future. These spaces were later filled by scandium, gallium, germanium, and technetium.
Atomic Radius
Atoms do not have sharp boundaries because the electrons around nucleus are diffused like cloud. Because of this, it is not possible to define the exact size of an atom.
One way to describe atomic size is by looking at the distance between two atoms that are not bonded but touch each other. For example, in solid krypton, atoms come into contact without forming bonds. The distance between the nuclei of two krypton atoms can be calculated from the density of the solid. Half of this distance is taken as the atomic radius. This type of atomic radius is called the non-bonding atomic radius or the van der Waals radius. It represents the size of an atom when it is not bonded to any other atom. For instance, the distance between two krypton nuclei in the solid state is about 400 pm, so its van der Waals radius is approximately 200 pm.
Another type of atomic radius is the bonding atomic radius, also called the covalent radius. Its definition is slightly different for nonmetals and metals. In nonmetals, it is half the distance between the nuclei of two identical atoms bonded by a covalent bond. In metals, it is half the distance between the nuclei of two adjacent atoms in a metallic crystal. For example, in a bromine molecule (Br₂), the distance between the nuclei of two Br atoms is 228 pm. Thus, the covalent radius of bromine atom is half of the bond distance between two bromine atoms which is 114 pm.
Similarly, in a chlorine molecule (Cl₂), the distance between the two nuclei is 198 pm, which gives a covalent radius of 99 pm. By this method, atomic radii can be assigned to most elements that form covalent bonds or metallic crystals.
A more general term, atomic radius, refers to the average bonding radius obtained from many experimental measurements of elements and compounds. The atomic radius shows the size of an atom when it is bonded to another atom. It is always smaller than the van der Waals radius, because bonded atoms are pulled closer together, whereas non-bonded atoms remain farther apart.
The concept of atomic radius is also useful for estimating bond length. The bond length between two covalently bonded atoms can be approximated as the sum of their atomic radii. For example, in iodine monochloride (ICl), the atomic radius of iodine is 133 pm and that of chlorine is 99 pm. Adding these gives a predicted bond length of 232 pm. The experimentally measured bond length is 232.07 pm, which closely matches the calculated value.
Comparison of Atomic Radii
| Type of Radius | Definition | Example | Relative Size |
| Covalent Radius | Half the distance between nuclei of two bonded identical atoms | Br = 114 pm; Cl₂ = 99 pm | Smaller |
| Metallic Radius | Half the distance between nuclei of two adjacent metal atoms in a crystal | Na = 186 pm | Moderate |
| van der Waals Radius | Half the distance between nuclei of two non-bonded atoms in contact | Kr = 200 pm | Largest |
Periodic Trends
Trend Along Periods
When we travel along periods from left to right, atomic radius decreases. The reason is that nuclear charge (number of protons) increases which brings electrons closer to the nucleus.
Trend Down the Groups
Down the groups, atomic radius of elements increases because number of electronic shells increases which , in turn, increases shielding effect. Nuclear charge also increases but the electronic shell factor dominates it.
Difference Between Nuclear Charge and Effective Nuclear Charge
The electrons around the nucleus of an atom experience an attractive force from the positively charged protons inside the nucleus. At the same time, however, electrons also repel one another (especially inner shell electrons repel outer shell electrons) because they all carry a negative charge. This repulsion shields or screens the outer electrons from the full attractive force of the nucleus-also called shielding or screening effect. As a result, the outermost shell electrons do not feel the actual force of attraction from the nucleus. This actual and net positive charge felt by these electrons is called the effective nuclear charge.
Periodic Trends in Transition Metals
Going across a period in transition metals, the trends in atomic radii is different from that explained above (group IA to VIIA). In transition metals, there are two factors working at the same time.
One is that nuclear charge increases in the same way as above which pulls electrons closer to the nucleus, decreasing atomic radii. The other factor is that electrons added to the atoms go into inner shell, (n-1)d, rather than outermost shell , ns. These electrons cause shielding the outermost shell electrons from the nucleus, which might be expected to increase the atomic radius.
In reality, the atomic radii of transition metals decrease only slightly because the increase in nuclear charge is almost counterbalanced by the shielding effect of the additional inner-shell electrons. There are, however, a few exceptions to this trend, which are beyond the scope of this course.
Frequently Asked Questions
Q1: Why can’t we measure atomic radius exactly?
Because orbitals do not have fixed boundaries; they only give a probability of where electrons are likely to be found.
Q2: Which radius is larger — covalent or van der Waals?
The van der Waals radius is always larger, since non-bonded atoms are farther apart than bonded atoms.
Q3: Can bond length always be predicted by adding atomic radii?
Yes, approximately. The bond length is usually close to the sum of the radii, though actual experimental values may differ slightly.
Q4: Which elements usually have their covalent radii measured?
Mainly nonmetals and metals that form covalent bonds or metallic crystals. Noble gases rarely form covalent bonds, so their van der Waals radii are used instead.
Q5: Why is atomic radius important?
It helps us understand bond lengths, bond strengths, periodic trends, molecular size, and physical properties such as melting and boiling points.
Ionic Radius
Ionic radius is the distance between the nucleus and outermost electrons of an ion (cation and anion). The trend in ionic radii depends on two factors:
- Nuclear charge
- Repulsion among valence shell electrons
Periodic Trends Going Left to Right
Periodic trends in ionic radii are difficult compared to atomic radii. The reason is that, along a period from left to right, the elements from group I to IV form cations while those in group V to VII, elements form anions. Ionic radii of cations and anions need to be studied with different parameters: nuclear charge and repulsions among valence shell electrons.
Ionis radii of cations decrease from left to right going through group I to IV because nuclear charge increases. The number of valence shell electrons remain the same, so inter-electronic repulsions do not matter.
When we switch from group IV to group V along a period, the ionic radii increase because three electrons are added to the valence shell of group V elements which increases repulsion between newly added three electrons and already lying five outermost shell electrons. This repulsion increases the size of the anion. Moving across the period, the ionic radii of anions decrease through group V to VII because nuclear charge increases.
Electron Affinity
Electron affinity is the amount of energy released when an electron is added to the valence shell of an isolated gaseous atom.
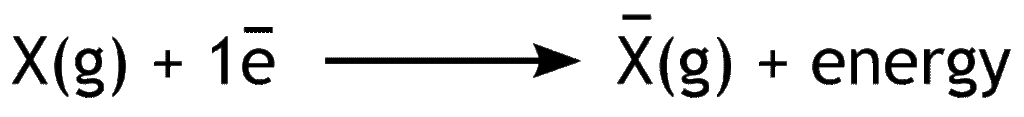
Electron affinity shows the measure of ability of a neutral atom to gain electron. Depending on the number of added electrons, there may be first electron affinity, second electron affinity, third electron affinity and so on.
The first electron affinity is negative, second and third electron affinities are positive. The positive values of second and third electron affinities is because electron is added to anions which repel the adding electron (like charges repel each other). To overcome this repulsion, energy is required, which makes the process endothermic and the electron affinities positive.
The electron affinities of non-metal atoms are negative (exothermic) because these atoms attract electrons to complete their outer shells, and this attraction results in the release of energy. However, the electron affinities of metals are positive (endothermic) because they do not need electrons, rather they want to lose electrons. When electron is added to the valence shell of an isolated metal atom, energy is required (endothermic process) because metal atoms repel the adding electrons.
Periodic Trends
Across a period from left to right, the electron affinities of elements increase, which means they become more negative.
