The term organic comes from a Greek word organikos which means “related to organ”. In 1808, the Swedish chemist Jons Jacob Berzelius proposed a theory known as the Vital Force Theory. According to this theory, organic compounds contained a spiritual force, called vital force, which is present in plants and animals. He believed that they could only be obtained from living organisms such as plants and animals.
Berzelius suggested that the formation of organic compounds required a spiritual “vital force” ( from the Latin word vitalis, which means “life”) present only in living organisms. He believed that this vital force was absent in non-living substances, and therefore organic compounds could not be prepared from inorganic materials in the laboratory.
Based on their origin, compounds were classified into two categories: organic compounds and inorganic compounds. Organic compounds were defined as substances derived from plants and animals, whereas inorganic compounds were obtained from non-living materials such as minerals and rocks.
In 1828, the German chemist Friedrich Wöhler rejected the vital force theory. He successfully synthesized the organic compound urea in the laboratory from the inorganic compound ammonium cyanate. This experiment proved that organic compounds could be prepared without involvement of any vital force. As a result, scientists redefined organic compounds based on their composition rather than their origin.
In 1859, the German chemist August Kekule defined organic compounds as compounds containing the element carbon. Similarly, organic chemistry was defined as the branch of chemistry that deals with the study of compounds containing carbon.
However, scientists later discovered that some carbon-containing substances are still classified as inorganic compounds. Examples include carbon dioxide (CO₂), carbon monoxide (CO), carbonates (CO₃²⁻), bicarbonates (HCO₃⁻), cyanides (CN⁻), and cyanates (CNO⁻). Because of these exceptions, the definition of organic compounds needed further changes.
Brain Boost ! Carbon dioxide and carbon monoxide are inorganic in nature because they come from volcanic activity and combustion. Similarly, carbonates and bicarbonates are inorganic in nature because they existed in the earth crust even before life existed.
Finally, in 1889, the German chemist Carl Schorlemmer proposed a clearer modern definition. He defined organic compounds as hydrocarbons and their derivatives. According to this modern definition of organic compounds, organic chemistry was defined as the branch of chemistry that deals with the study of hydrocarbons and their derivatives.
Brain Check ! Organic compounds generally have carbon-carbon or carbon-hydrogen bonds in their molecules.
Why Carbon so Important?
Carbon is the central element in organic chemistry because it has unique properties. Due to these properties, carbon can alone form compounds greater in number than all other elements in nature can form. Carbon stands unique among all elements in the universe due to the following properties:
i. Tetravalency
Carbon has four electrons in its valence shell (1s2 2s2 2p2). It uses all four electrons for bonding to form four covalent bonds with other atoms. This property allows carbon to act as a branching point and forms three dimensional and tetrahedral complex structures, not just simple straight molecules.
ii. Catenation
Catenation (from the Latin word catena, meaning “chain”) is the ability of carbon to form bonds with its own atoms, making long chains and rings. This occurs because carbon forms strong carbon–carbon bonds due to its small atomic size. As a result, carbon can form many different structures, which explains the large number of organic compounds.
Brain Box Carbon can form many different organic compounds even when a fixed number of carbon atoms combine with a fixed number of atoms of other elements. For example, decane (C10H22) can form 75 isomers and eicosane (C20H42) is able to form 350 000 isomers.
iii. Multiple Bond Formation
Carbon can form double and triple bonds with other carbon atoms and with elements like oxygen and nitrogen. Its small atomic size allows the atoms to come close together, making it possible to form π (pi) bonds. Therefore, carbon can form stable double and triple bonds. In contrast, other elements in the same group, such as silicon, usually do not form stable double and triple bonds because their atoms are larger in size.
Did You Know? By early 2026, the total number of compounds (both organic and inorganic) registered by the Chemical Abstracts Service (CAS), a division of the American Chemical Society (ACS), is approximately 204 million, of which nearly 180 million are organic compounds.
Importance of Catenation in Organic Chemistry
Catenation is unique ability of carbon to make strong bonds with its own atoms and make long chains and rings. This property of carbon makes organic chemistry a separate and vast branch of chemistry. Catenation plays an important role in the following areas of organic chemistry:
i. Structural Diversity Catenation allows carbon to act as a backbone of long straight chains, branched chains and rings. This is the reason why we have a big variety of molecular shapes.
ii. Molecular Complexity Carbon can bond its own atoms with one another and make molecules ranging from very simple molecules (methane) to very complex molecules (proteins and carbohydrates).
iii. Isomerism The ability of carbon to link in different ways gives different compounds with the same set of atoms. Carbon atoms catenate in different patterns, so this property increases the number of organic compounds.
iv. Basic Life Life depends on large molecules, like proteins, carbohydrates, and fats. Catenation of carbon is the only reason why these life-making molecules exist.
Representation of Organic Molecules
Some organic compounds have very complex structures, so they need more than one different types of formula to represent them.
A general formula is an algebraic expression used to represent a homologous series (family) of organic compounds. Each family of organic compounds has its own characteristic general formula.
i. General Formula.
A general formula helps us predict the molecular formula of any member of that family by substituting different values of n (number of carbon atoms) so it is also named as general molecular formula of a family. For example, the general formulae of some important homologous series are given in the table below.
| Homologous Series | Alkanes | Alkenes | Alkynes | Alcohols | Aldehydes/Ketones | Carboxylic acid |
| General Formula | CnH2n+2 | CnH2n | CnH2n-2 | CnH2n+2O | CnH2nO | CnH2nO2 |
Remember that aldehydes and ketones have the same general formula CnH2nO but they differ in their functional groups.
ii. Molecular Formula.
A molecular formula shows the number and types of atoms in a molecule. It tells you nothing about how the atoms are connected in a molecule. The molecular formulas of some molecules are given bel.
For example, C2H6, C3H6, C2H2, C2H6, C2H6O, C2H4O2 are molecular formulas of ethane, propene, ethyne, ethanol, and ethanoic acid respectively.
iii. Structural Formula
A structural formula represents the connection between atoms in a molecule. There are three types of structural formulas.
a. Condensed Formula
A condensed formula shows how atoms are bonded in a molecule, without drawing single bonds. The hydrogen atoms are grouped with the carbon atoms to which they are bonded. The substituents/branches are put in parentheses.
Examples, CH3CH2CH2CH(CH3)CH2CH3 3-methylhexane
CH2=CHCH2CH3 but-1-ene
b. Displayed (Full Structural) Formula
Displayed formula is a graphic representation that shows all atoms and all bonds between atoms in a molecule. This formula can easily differentiate between the structural isomers for a given molecular formula. The displayed formulas of some compounds are given below.
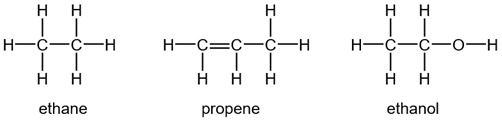
Mostly, students forget drawing bond between hydrogen and oxygen in hydroxyl group of alcohols.
c. Skeletal Formula
The skeletal formula (also called the line-angle formula) is a simplified representation of organic molecules drawn in a zig-zag manner, in which carbon and hydrogen atoms are not shown. Each end and corner of a line represents a carbon atom. The term skeleton is used because it deals with the carbon framework (backbone) of molecules.
Hydrogen atoms attached to carbon are omitted but are assumed to be present to satisfy the tetravalency of carbon (each carbon forms four bonds). However, heteroatoms such as N, O, Cl, S, etc., and any hydrogen atoms attached to them are shown clearly in the skeletal formula. Some example of skeletal formula are given.

Single and double-bonded carbon atoms are represented in a zig-zag pattern because their respective bond angles,109.5o for sp3 (tetrahedral) and 120o for sp2 (trigonal planar), create natural bends in the chain. In contrast, triple bonds are drawn in a straight line because sp hybridized carbons possess a linear geometry with a bond angle of exactly 180o.
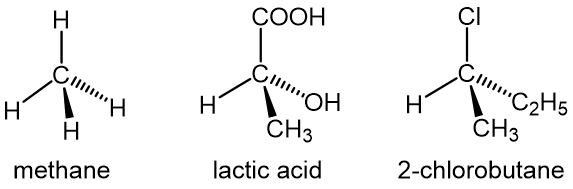
d. Stereochemical Formula (3D Formula)
Stereochemical formula (also called wedge-dash formula) is advanced version of structural formula of organic compounds showing all atoms and bonds in three-dimension. It shows the direction in which atoms and bonds are oriented. Some examples of stereochemical formula are given below:
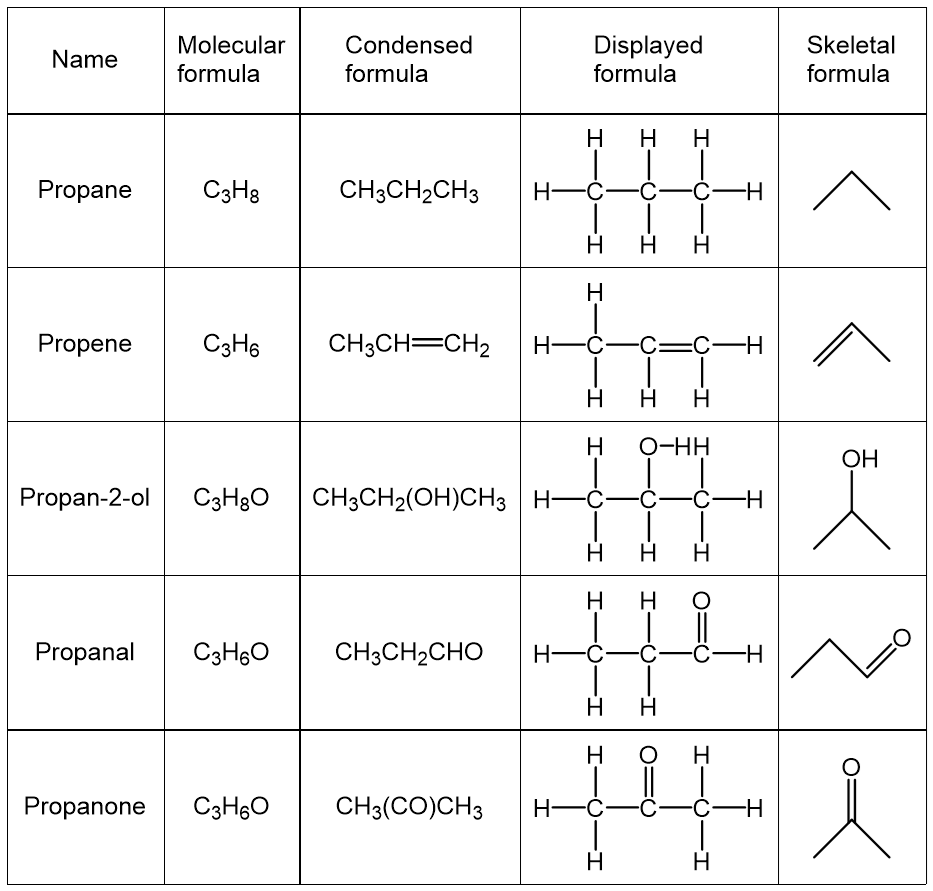
The following table shows molecular formulas, condensed formulas, displayed formulas, and skeletal formulas of some important organic compounds.
Functional Group
An atom or group of atoms that gives characteristic chemical and physical properties to organic compounds is called functional group.
All organic compounds consist of two main parts: the carbon skeleton and the functional group. The carbon skeleton forms the basic framework of the molecule and is generally less reactive. On the other hand, the functional group is the reactive part (active site) of the molecule and is responsible for its characteristic properties. For example, alcohols are represented by the general structural formula ROH, where the alkyl part (R) represents the relatively unreactive carbon skeleton, while the hydroxyl group (–OH) is the reactive part known as the functional group of alcohols.
Remember ! Alkanes have no functional group because they lack heteroatoms (like O, N, or S) or pi (π) bonds. They have strong and non-polar C-C and C-H sigma (δ) bonds which means that they do not possess a specific reactive site that can easily initiate a chemical reaction.
The physical properties of organic compounds, such as melting point and boiling point, largely depend on the nature of the functional group present in the molecule. Therefore, functional groups play a major role in determining the physical properties of organic compounds.
Similarly, most chemical reactions of organic compounds occur at the functional group. Hence, the functional group is responsible for determining the chemical properties and reactivity of organic compounds.
Anology ! The pizza base (bread and sauce) represents the carbon skeleton of an organic compound. The toppings (such as vegetables, meat, or cheese) represent the functional group. The flavour or taste of the pizza represents the chemical and physical properties of the compound.
It is important to note that double bonds (C=C) and triple bonds (C≡C) are also considered functional groups, as they influence both the physical and chemical properties of organic compounds.
Do You Know? Alkanes have no functional group because they lack heteroatoms (like O, N, or S) or pi (π) bonds. They have strong and non-polar C-C and C-H sigma (δ) bonds which means that they do not possess a specific reactive site that can easily initiate a chemical reaction.
Homologous Series
A homologous series is a group of organic compounds which have same functional group, where each successive member differs by one methylene group (-CH2-). There are nearly 180 million organic compounds, so it is practically very difficult to study them individually. To make their study easy, they have been classified into a few homologous series. The following table shows important functional groups and corresponding homologous groups.
